Health and Tech: MCED tests to help detect cancers early
MCED tests Utilise blood, urine samples to detect cancer
Published Date - 30 March 2023, 12:45 AM
Undergoing tests for cancer diagnosis is quite traumatic for patients, as they not only have to prepare mentally and physically to undergo an invasive procedure for submitting tissue samples for biopsy but also experience stress and anxiety between the time when tests are done and the results arrive.
Difficult diagnostics are also a barrier that de-motivates people from getting the tests done on time.
In the last few years, however, independent scientific groups and pharma companies worldwide have been trying to address such issues in cancer diagnosis by developing early cancer tests that are less traumatic, non-invasive, quick, and accurate.
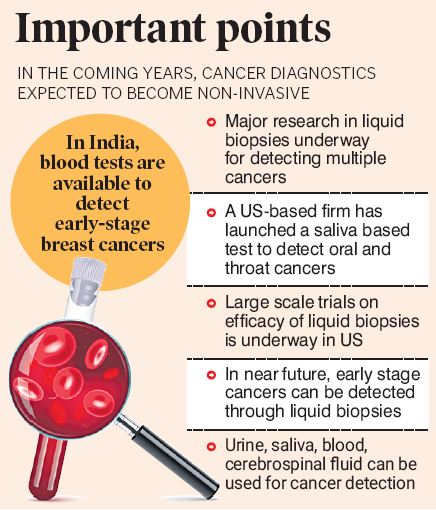
While we are still a few years away from a definitive non-invasive cancer diagnostic test, cutting edge research is underway in several countries, especially United States where scientists are developing Multi-Cancer Early Detection (MCED) tests that utilise blood or urine samples to detect multiple cancers.
Liquid biopsies or MCEDs
National Cancer Institute (NCI) of the United States, which is involved in developing such tests, has defined liquid biopsies as laboratory tests done on a sample of blood, urine, or other body fluid to look for cancer cells from a tumor or small pieces of DNA, RNA, or other molecules released by tumor cells into a person’s body fluids.
A liquid biopsy allows multiple samples to be taken over time, which may help doctors understand what kind of genetic or molecular changes are taking place in a tumor. A liquid biopsy may be used to help find cancer at an early stage. It may also be used to help plan treatment or to find out how well treatment is working or if cancer has come back.
According to MIT Technology Reviewer, liquid biopsies (blood/urine) work by looking for remnants of tumor cells that are left behind after the immune system attacks them. The main principle behind such tests is that the remnants or debris of dead tumors turn up in the bloodstream, which when collected and tested could warn of cancer at an early stage.
Saliva Tests:
Several pharma companies are involved in developing simple saliva tests to accurately detect certain cancers. In 2022, the USFDA (US Food and Drug Administration) had given a green signal for the launch of a saliva-based detection test for the diagnosis of oral and throat cancers.
The saliva oral and throat cancer tests, developed by US company Viome, are home-based screening tools that are based on AI and machine learning. The tool analyses a saliva sample for the activity genes that belong to bacteria in the mouth. When individuals develop tumors on the lips, tongue, or throat, the bacterial communities in the mouth get modified and the test is able to detect the modification.
Although traditional cancer diagnosis techniques like tissue biopsy, mammography, pap-smear, cervical cancer screening are considered gold standards, in the coming years, physicians might not need scalpels, needles or endoscopy because of the promise of liquid biopsies that are conducted through urine, blood, saliva, cerebrospinal fluid, and semen.